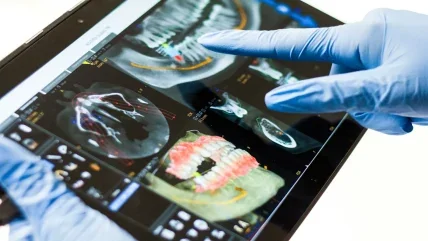
DentalMonitoring said its DentalMonitoring Software has received certification under the European Union Medical Device Regulation 2017/745 (EU MDR) and meets the criteria for CE marking as a class IIa medical device.
France-based DentalMonitoring is an artificial intelligence (AI)-based remote monitoring firm focused on orthodontic care.
Its DentalMonitoring solution is a medical device software designed to assist in remote monitoring of dental treatments, orthodontic treatments, oral health, and treatment progress.
It uses image processing algorithms to analyse high-resolution intraoral scans captured by patients using the DM App, alongside proprietary hardware, and smartphones.
The software also supports clinicians in diagnosis and treatment planning through regular clinical updates.
Last month, the orthodontic care company received De Novo approval from the US Food and Drug Administration (FDA) for the DentalMonitoring solution.
The EU MDR, which supersedes the Medical Device Directive 93/42/EEC (MDD), imposes more stringent requirements for clinical evaluation, risk management, post-market surveillance, and data collection on medical devices.
DentalMonitoring has achieved this milestone four years ahead of the deadline.
The company said clinicians using the medical device software will gain enhanced visibility into treatment progress, while patients benefit from greater convenience.
Automated notifications and in-app communication with practice staff will improve patient accountability and foster better communication between patients and healthcare providers.
According to the French firm, this enhanced connectivity helps promote patient adherence to orthodontic treatment protocols.
DentalMonitoring CEO and co-founder Philippe Salah said: “I’m incredibly proud that DentalMonitoring has achieved EU MDR certification along with US/FDA DeNovo 510k clearance.
“These achievements celebrate our relentless pursuit of innovation and excellence in oral care. Our AI-powered solutions are now recognised at the highest regulatory levels, enabling us to expand our reach and impact more lives with advanced dental technology.”






