All articles by Staff Writer
Novo Nordisk acquires Embark Biotech, including its lead asset targeting obesity and other cardiometabolic diseases.
Embark Laboratories announced today that Novo Nordisk has acquired Embark Biotech, including its lead metabolic program, and has entered a three-year research and development collaboration to discover and develop novel pharmaceuticals to treat obesity and related co-morbidities.
Casio to Release Uterocervical Observation Camera and Camera Stand in Europe
Completion of the Declarations of Conformity to European Medical Device Regulations (EU MDR) and UK Medical Device Regulations (UK MDR)
Leslie McDonnell joins Mölnlycke’s Board of Directors
Leslie McDonnell has been elected Member of Mölnlycke AB’s Board of Directors on 1 September 2023.
Baltimore VA Medical Centre opts for CareView patient monitoring tech
This system provides real-time insights to healthcare providers for predicting inpatient falls.
Roche’s Alecensa delivers unprecedented Phase III results for people with ALK-positive early-stage lung cancer
Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that the Phase III ALINA study evaluating Alecensa® (alectinib), compared with platinum-based chemotherapy, met its primary endpoint of disease-free survival (DFS) at a prespecified interim analysis. Alecensa demonstrated a statistically significant and clinically meaningful improvement in DFS as adjuvant therapy in people with completely resected stage IB (tumour ≥4cm) to IIIA (UICC/AJCC 7th edition) anaplastic lymphoma kinase (ALK)-positive non-small cell lung cancer (NSCLC). Alecensa is the first and only ALK inhibitor to demonstrate a reduction in the risk of disease recurrence or death for people with early-stage ALK-positive NSCLC in a Phase III trial.
Healthcare companies using Google Cloud generative AI tools
Huma said it expects the technology to automate its clinical summary reports and improve its data triaging processes.
Start-up Time is Brain monitors brain function after stroke
Interview with Alicia Martínez (PhD), CEO, Time is Brain
MEDICA 2023 + COMPAMED 2023: “Where Healthcare is going” – the leading motif is reflected in a wide variety of formats
The entire value chain of medical care, product development and manufacture in focus at one location
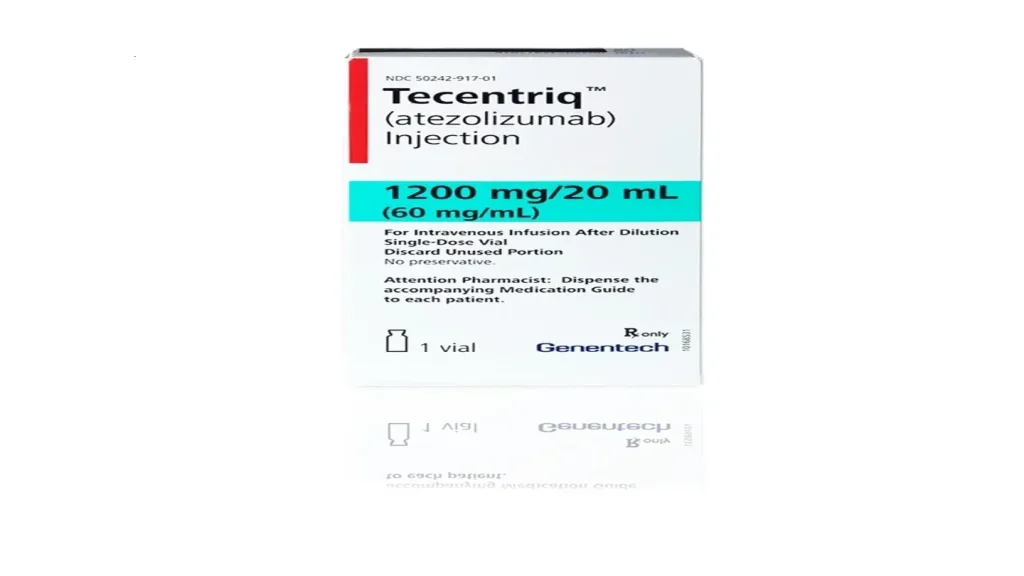
Roche’s Tecentriq becomes the first subcutaneous anti-PD-(L)1 cancer immunotherapy available to patients in Great Britain, reducing treatment time to just minutes
Roche (SIX: RO, ROG; OTCQX: RHHBY) announced today that Tecentriq® SC (atezolizumab) has been approved by the Medicines and Healthcare products Regulatory Agency (MHRA) in Great Britain. It will be provided by the National Health Service (NHS) England. Injecting Tecentriq subcutaneously (under the skin) takes approximately seven minutes, compared with 30-60 minutes for intravenous (IV) infusion. Tecentriq SC will be available to patients in Great Britain for all indications in which the IV formulation of Tecentriq has been previously approved, including certain types of lung, bladder, breast and liver cancer.2
Trialogics Opens London Office, Advancing Remote Device Management Services for Clinical Trials
Trialogics, a leading clinical trial software and services company, providing eConsent, IRT (RTSM), and eCOA, announced today that it is growing its overall services by expanding their Remote Device Management capabilities.